Against a backdrop of rising global obesity, researchers trace brown fat to an unexpected embryonic source.
Krems (Austria), 12. March 2026 – A team of scientists has identified an unexpected embryonic “starting point” for several brown-fat depots: a small cell niche around the dorsal aorta. Brown adipose tissue helps mammals to regulate their body temperature, and it is drawing growing attention as obesity rises worldwide. Using a combination of time-controlled genetic lineage tracing and single-cell RNA sequencing in early mouse embryos, the researchers show that progenitor cells from this aorta-adjacent region later contribute to brown fat in multiple body locations – while the best-known depot between the shoulder blades (interscapular region) is only minimally derived from this source. The work was carried out primarily at several top research facilities in Berlin and parts of it are now brought to Karl Landsteiner University (KL Krems) through the senior scientist Prof. Sigmar Stricker.
Brown adipose tissue – often called ‘brown fat’ – is not uniform across the body: it appears in distinct anatomical depots, and these depots differ in cellular composition and gene activity. This matters because researchers often use one depot as a model for “brown fat” in general. If depots do not share the same developmental origin, they may also differ in how they are regulated – and in how reliably findings from one region translate to another. The new study tackles this uncertainty by mapping exactly which embryonic progenitor pools feed into which depots, and when those fate decisions are made.
Hidden Location
“Previous studies suggested that brown fat depots might not all develop in the same way,” says Prof. Sigmar Stricker, Head of the Department of Cell Biology at KL Krems. “We now used lineage tracing and single-cell data to check this idea in more detail – and, indeed, we found strong evidence for a yet unknown location of progenitor cells of brown adipose tissue near the dorsal aorta.”
The team around Prof. Stricker focused on cells expressing the gene Osr1 (a transcription factor) and followed the development and migration of these cells over time. By labelling Osr1-positive cells at defined embryonic stages, they uncovered clear depot-specific patterns: Osr1-lineage cells contributed strongly to brown fat beneath the shoulder blade (subscapular) and in the neck (cervical) regions, but only minimally to the large depot between the shoulder blades (interscapular) – a widely used reference tissue in mouse studies. An even earlier labeling time point still revealed substantial contributions to the subscapular and cervical depots, indicating that relevant Osr1-positive progenitors are present very early in development.
Single-cell RNA sequencing added the molecular context. At a later embryonic stage, Osr1-positive cells showed a clearer “pre–brown fat” signature. At an earlier stage, however, a subset displayed a broader, multipotent profile, with gene-expression features linked to vascular- and muscle-related programs. Consistent with this, lineage tracing indicated that early Osr1-positive cells can contribute not only to adipose tissue, but also to other mesoderm-derived tissues in defined embryonic regions.
From Aorta to Adipose
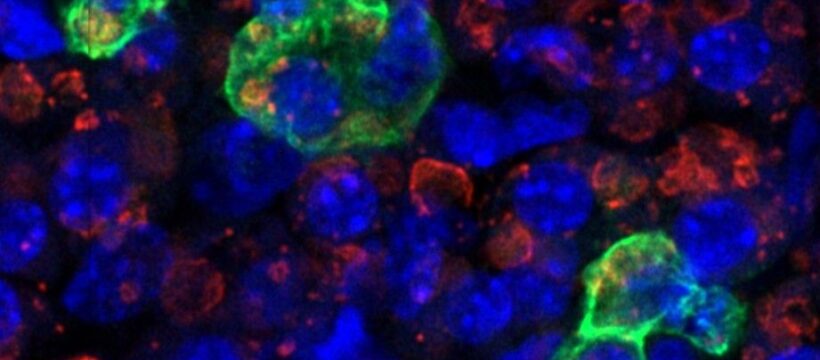
The anatomical clue to the origin and route of the progenitor cells emerged when the researchers examined embryos at the earlier stage using immunofluorescence: Osr1-positive cells were enriched in the dorsal aortic compartment, a narrow zone around the embryonic aorta. Over subsequent days, time-resolved tracing suggested that Osr1-lineage cells disperse from this area toward more back-and-side regions of the embryo (dorso-lateral territories). This pattern is consistent with ideas about mesoangioblasts – vessel-associated, multipotent progenitors. The authors therefore propose that dorsal aorta–associated, Osr1-positive cells represent an in vivo source of progenitors that can later form several brown-fat depots.
A substantial part of the experimental work and infrastructure for this study was based at Freie Universität Berlin, with additional expertise coming from the Max Delbrück Center Berlin, the Max Planck Institute for Molecular Genetics in Berlin, the Charité University Medicine Berlin, and the German Institute of Human Nutrition (DIfE), besides the KL. Prof. Stricker’s current base at Karl Landsteiner University connects these findings to the university’s broader research agenda and enables follow-up work at the interface of developmental biology and metabolic health.
Original publication: The dorsal aortic compartment is a developmental source of brown adipose tissue in mice. S. Heider, C. Fischer, A. K. Secener, P. Vallecillo-García, G. Kotsaris, Z. G. Meisen V. Pawolski. C. Giesecke-Thiel, T. Conrad, T. J. Schulz, S. Sauer, S. Stricker. Nature Communications (2026) 17:286, doi:10.1038/s41467-025-68147-9. https://kris.kl.ac.at/en/publications/the-dorsal-aortic-compartment-is-a-developmental-source-of-brown-/
More on KL Krems research: https://www.kl.ac.at/en/research/research-blog
Karl Landsteiner University (03/2026)
The Karl Landsteiner University (KL Krems) is an internationally recognized educational and research institution located on the Campus Krems. KL Krems offers modern, demand-oriented education and continuing education in medicine and psychology as well as a PhD programme in Mental Health and Neuroscience. The flexible educational programme is tailored to the needs of students, the requirements of the labour market and the challenges of science. The three university hospitals in Krems, St. Pölten and Tulln and the MedAustron Ion Therapy and Research Centre in Wiener Neustadt guarantee clinical teaching and research of the highest quality. In its research, KL Krems focuses on interdisciplinary fields with high relevance to health policy – including mental health and neuroscience, molecular oncology as well as the topic of water quality and the associated health aspects. KL Krems was founded in 2013 and accredited by the Austrian Agency for Quality Assurance and Accreditation (AQ Austria).
Scientific Contact
Prof. Sigmar Stricker
Faculty of Medicine
Karl Landsteiner University
Dr.-Karl-Dorrek-Straße 30
3500 Krems / Österreich
T+43 2732 7209 0761
E sigmar.stricker@kl.ac.at
Karl Landsteiner University
Mag. Selma Vrazalica, BA
Communication, PR & Marketing
Dr.-Karl-Dorrek-Straße 30
3500 Krems / Austria
T +43 2732 72090 237
M +43 664 883 99 603
Copy Editing & Distribution
PR&D – Public Relations for Research & Education
Dr. Barbara Bauder-Jelitto
Kollersteig 68
3400 Klosterneuburg / Austria
M +43 664 1576 350
L https://www.linkedin.com/company/prd-public-relations-für-forschung-bildung